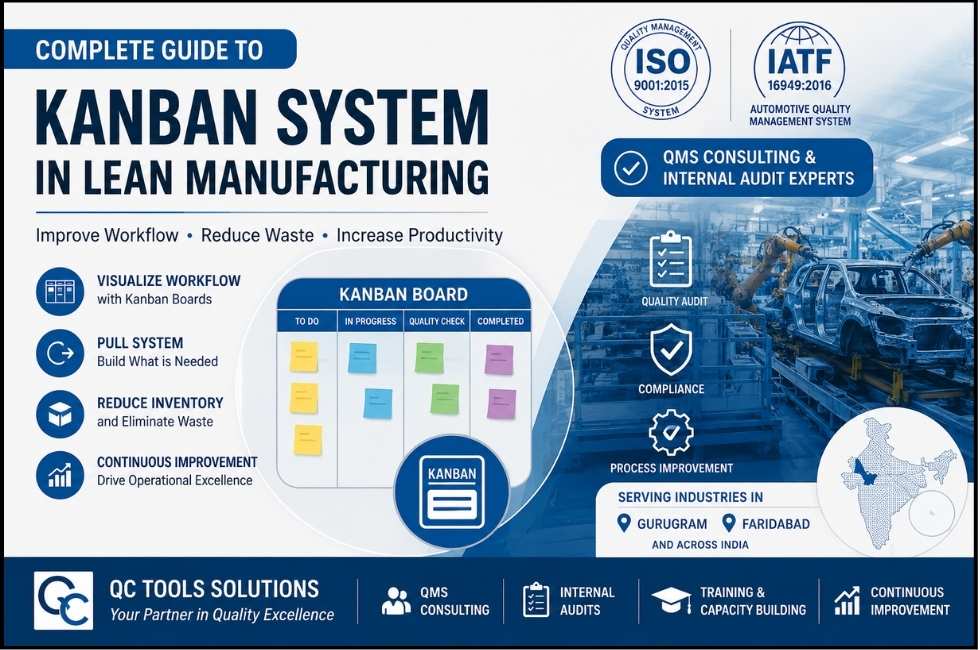
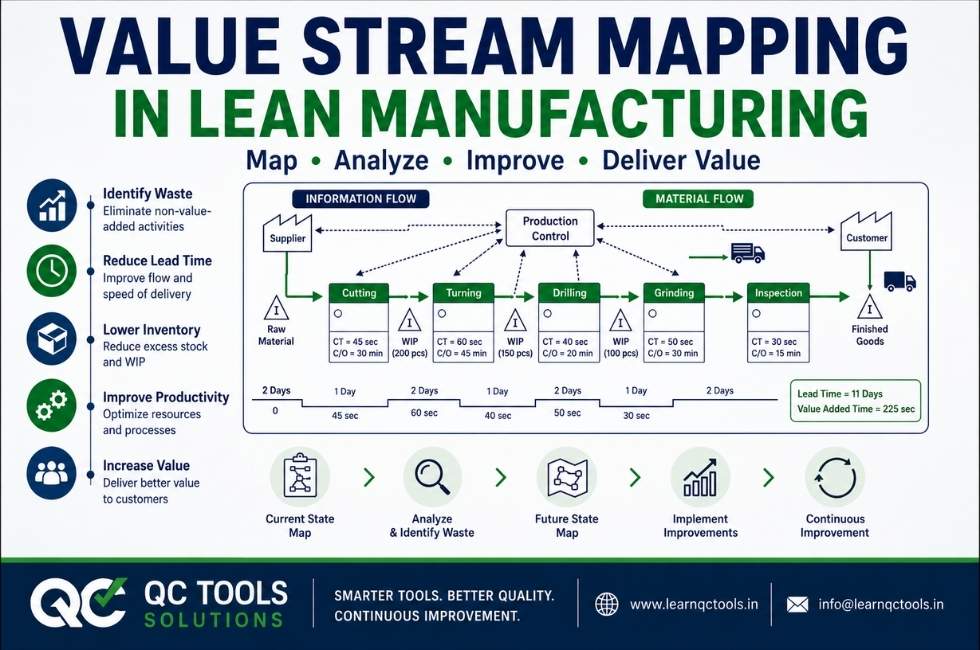
Complete IATF 16949 Non-Conformities (NC) Guide: Types, Examples & CARA
To close IATF non-conformities received from the IATF auditor, upload the JSON file in the NC Cara portal. CARA in IATF is the Common Audit Report Application. This is the application on the IATF Global oversight website. There are 7 steps for closing the IATF non-conformity on the NC CARA portal. 7 steps to close … Read more